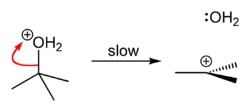
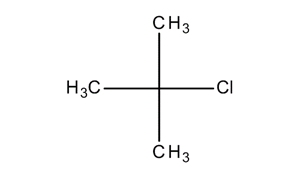
organic chemistry - Why is the reaction rate of tert-butyl chloride solvolysis faster in 80% water/20% acetone, than in 70% water/30% acetone? - Chemistry Stack Exchange
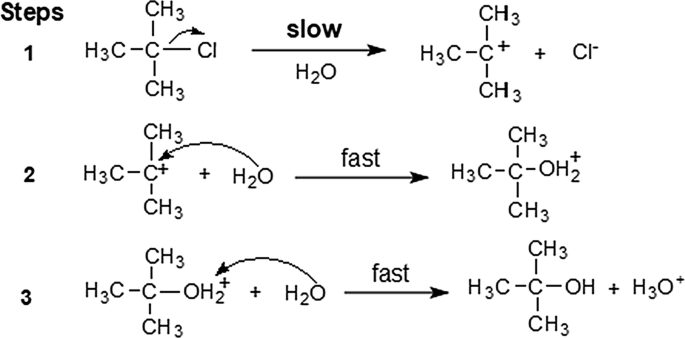
A solvent-catalyzed four-molecular two-path solvolysis mechanism of t-butyl chloride or bromide in water or alcohol derived by density functional theory calculation and confirmed by high-resolution electrospray ionization-mass spectrometry | SpringerLink

What is order reactivity of SN1 and SN2 of n-butyl chloride, n-butyl bromide, sec-butyl chloride, tert-butyl chloride, and crotyl chloride. Why? | Homework.Study.com

A solvent-catalyzed four-molecular two-path solvolysis mechanism of t-butyl chloride or bromide in water or alcohol derived by density functional theory calculation and confirmed by high-resolution electrospray ionization-mass spectrometry | SpringerLink